Examples of potential side effects / adverse events include:
- Headache
- Nausea (feeling sick)
- Vomiting
Miguel is attending a regional medical conference. One of the speakers at the conference is a patient who is receiving a Roche medicine for her condition. During the presentation, the patient tells the audience that when she first started taking the medicine she experienced frequent headaches. Because of this, her doctor reduced his dosage and after that, her headaches went away.
Miguel must report this potential side effect because the new information may impact the safety profile of the medicine.
An Adverse Event (AE), also known as a side effect, is an unwanted and unintended response in a patient, after receiving a medicinal product, that may or may not have been caused by treatment with this product. This includes all events where harm or unintended and unwanted response in a patient, user or other person as a result of the use of a medical device. The information you provide helps to ensure the safety of our products and our patients.
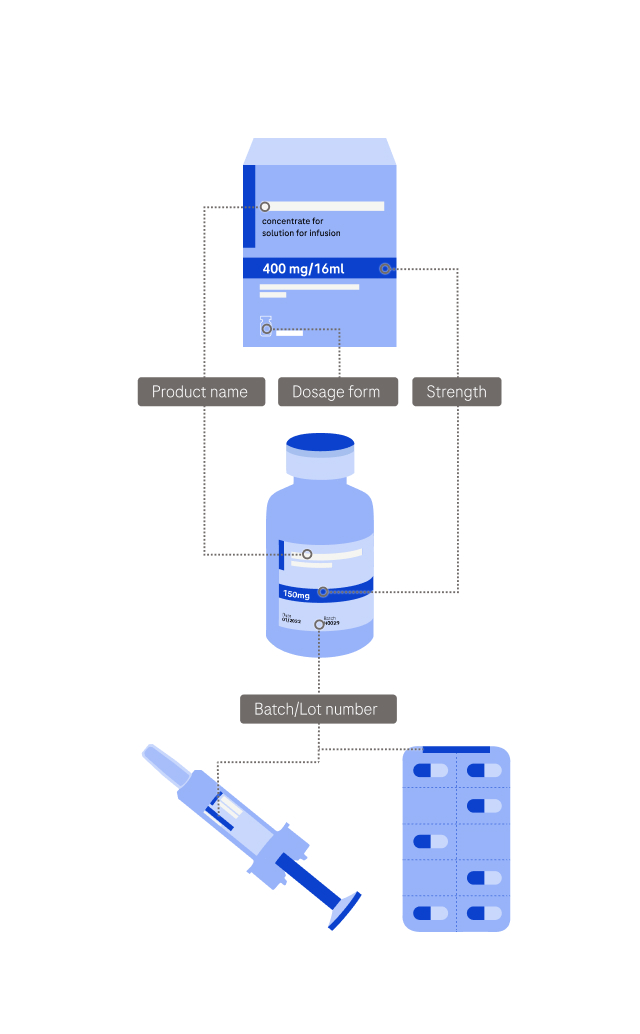
Side effects may include (but are not limited to) symptoms like:
Please describe the adverse event as accurate as possible and provide as many details as possible, like for example: The [side effect] occured after taking [the product] on [date]. The [side effect] started on [date] and the patient was admitted into hospital. The [side effect] stopped on [date].
The pregnancy-related questions are meant to collect information about side effects and pregnancy. Thank you for contributing to making our products safer.
Describe here the side effect, if this has resolved or is ongoing, how serious this is/was, the outcome and any further information that may be relevant both before, during and after the side effect occurred. For medical devices please also describe the way the device caused or contributed to the event and if relevant clearly describe the device malfunction causing the event.
Information about the use of our medicines during pregnancy is valuable to us to increase knowledge of their safety and inform patients. Please note, patients should consult with their healthcare professional to be informed on the safety of taking a medicine during pregnancy.
To improve the safety of our medication, any information (at least 1 identifying detail) you can provide is very helpful. The data will be stored in accordance with data privacy regulations.
If you consent in contacting your healthcare professional, please provide details below.
I consent to my data being processed for the purpose of responding to my inquiry and in accordance with the Roche Privacy Policy & Privacy Notice for Pharmacovigilance.
You consent, by ticking the box above, to the processing of your data for the purposes of responding to your inquiry and in accordance with the Roche Privacy Notice for Pharmacovigilance, medical information and product complaints. If you do not consent to the data processing, we will not be able to reply to your message and kindly ask you to use other channels to contact us.
Roche (Philippines) Inc. ("Roche") is required by law to record and/or report adverse events. Therefore, your data will be processed for these purposes in accordance with the specific GVP (pharmacovigilance) legislation, as further described in the Roche Privacy Notice for Pharmacovigilance, Medical Information and Product Complaints.
Roche will retain the personal information you provide for the purposes of responding to your request, following up on such requests, retaining the information in Roche's global pharmacovigilance database for reference purposes and to comply with our legal and regulatory record-keeping and reporting obligations. The retention period of the data is in accordance with the relevant laws and regulations.
By including personal data of another data subject in the form, you declare the lawfulness of the processing of this personal data.
The Privacy Policy provides more detailed information about your rights and how Roche processes personal data.
By following this link, you are leaving Medical Information and entering a website that is not owned or controlled by Roche. Roche does not take any responsibility for access to or use of this website, nor for any content therein.